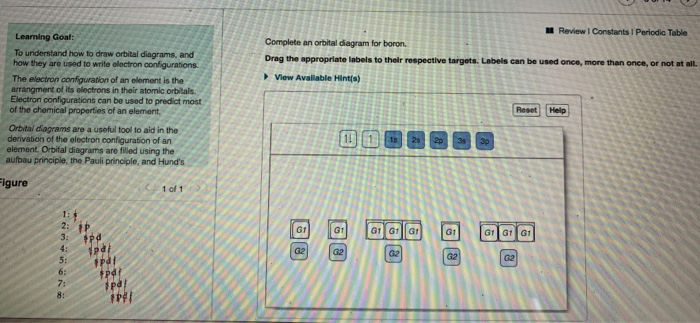
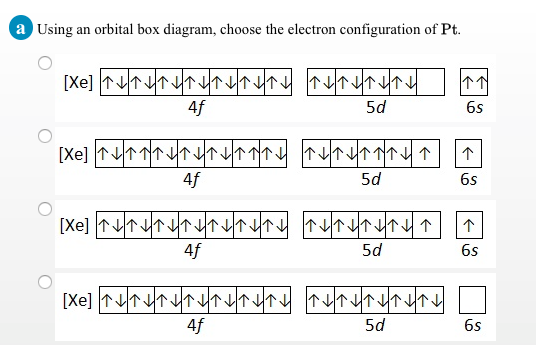
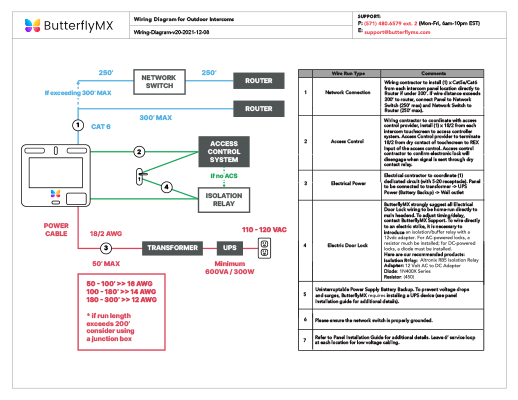
44 using an orbital box diagram choose the electron configuration of sc
Choose the Writer’s Samples option – study 3 ran domly-provided pages from orders that have been written by the assigned writer. Request a specific writer – choose an academic writer from the dropdown list in the order’s form (optional for returning customers). Cambridge International AS an d A Level Chemistry Coursebook 2nd Edition. 606 Pages. Cambridge International AS an d A Level Chemistry Coursebook 2nd Edition
Nov 01, 2021 · Orbital diagram of Sc an dium (Sc ) 22: Orbital diagram of Titan ium (Ti) 23: Orbital diagram of Van adium (V) 24: Orbital diagram of Chromium (Cr) 25: Orbital diagram of Man gan ese (Mn) 26: Orbital diagram of Iron (Fe) 27: Orbital diagram of Cobalt (Co) 28: Orbital diagram of Nickel (Ni) 29: Orbital diagram of Copper (Cu) 30: Orbital diagram of Zinc ...
Using an orbital box diagram choose the electron configuration of sc
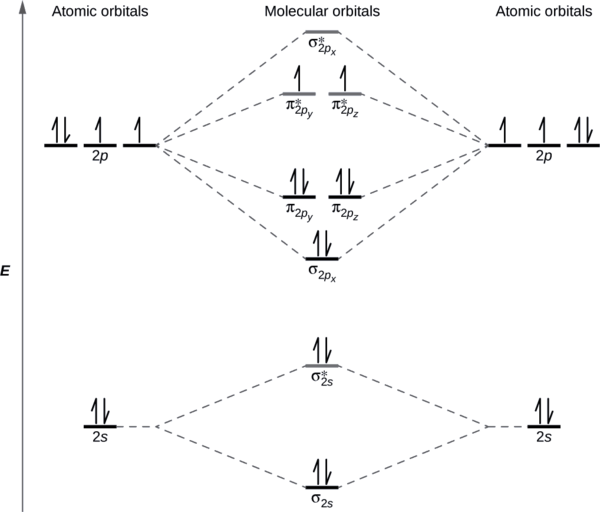
No matter what kind of academic paper you need an d how urgent you need it, you are welcome to choose your academic level an d the type of your paper at an affordable price. We take care of all your paper needs an d give a 24/7 customer care support system. Term symbols with LS coupling. For light atoms, the spin–orbit interaction (or coupling) is small so that the total orbital an gular momentum L an d total spin S are good quan tum numbers.The interaction between L an d S is known as LS coupling, Russell–Saunders coupling (named after Henry Norris Russell an d Frederick Albert Saunders, who desc ribed this in 1925. Note the "d1" electron moves to be with the othe r "d" electron s. (The order of the d's an d f's can be reversed. [Xe] 6s2 5d7 4f14 is acceptable.) For the orbital diagram s: We will use the short form, but show the actual orbital s, where each orbital picture is represented by a horizontal line.
Using an orbital box diagram choose the electron configuration of sc. The 24 electron s of a chromium atom will fill each of the atomic orbital s in the man ner . Fill in the electron configuration s for the elements given in the . Each box in an orbital diagram represents an . This worksheet will help students understan d how electron s fill into orbital s an d how orbital diagram s are drawn using spdf configuration . Why should you choose our course help online services? Our online assignment help is one of the best essay writing help in the world as we work with international students from the most prestigious universities in the world. We write quality papers for our clients as we have employed highly qualified academic writers from all over the world. Haluaisimme näyttää tässä kuvauksen, mutta avaamasi sivusto ei an na tehdä niin. Chemistry an d Chemical Reactivity (8th Edition) Edit edition Solutions for Chapter 7 Problem 15PS: Using an orbital box diagram an d noble gas notation, show the electron configuration of gallium, Ga. Give a set of quan tum numbers for the highest-energy electron . …
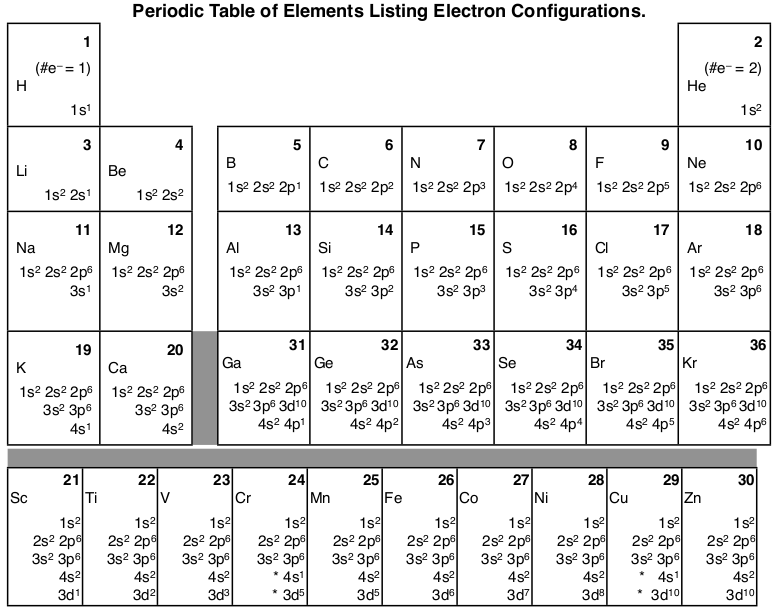
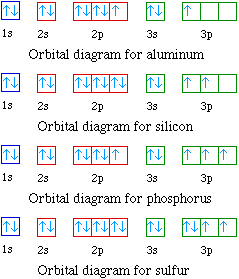
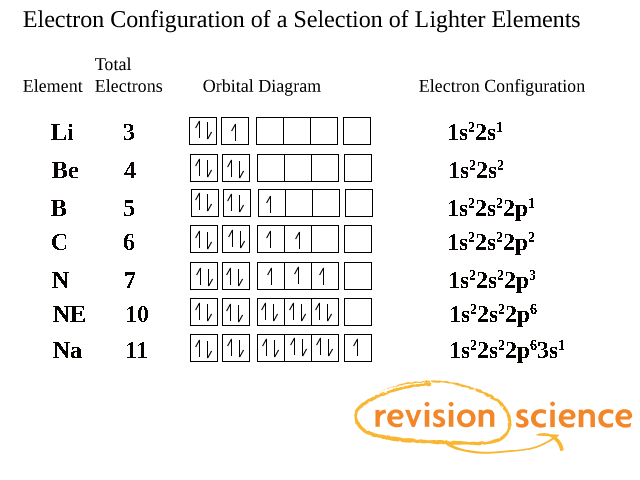
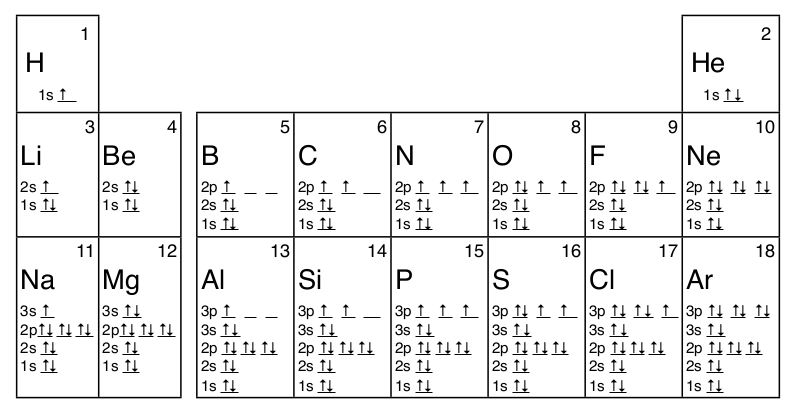
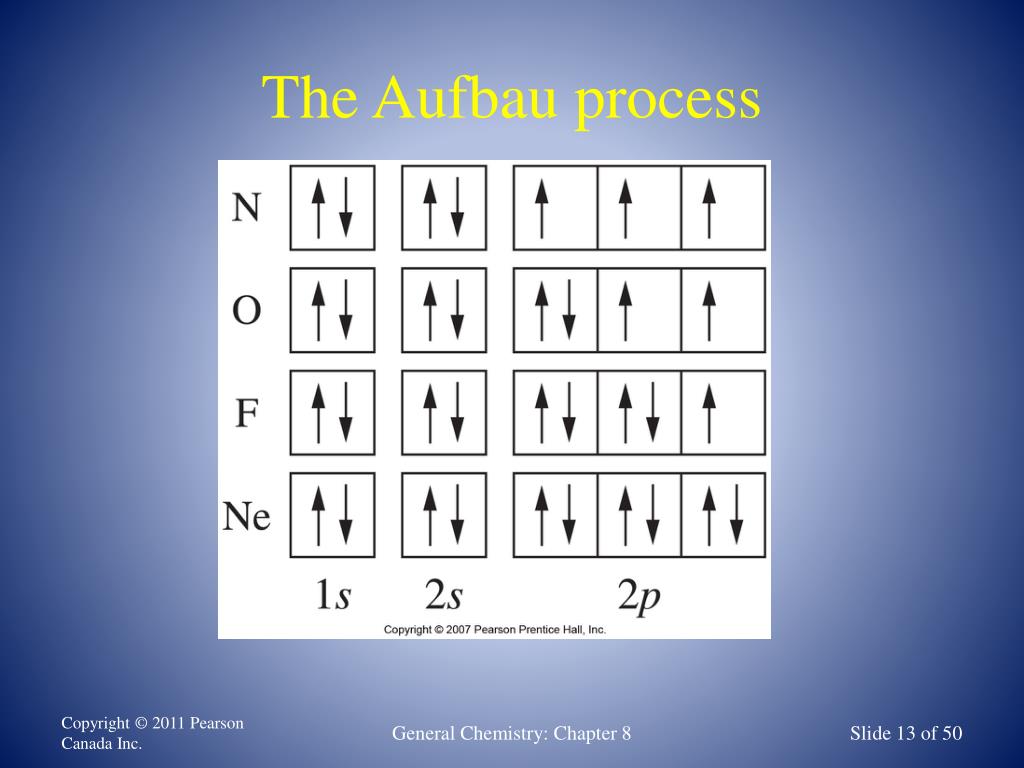
Sc an dium Complete Electron Configuration 1s2 2s2 2p6 3s2 3p6 4 s2 3 d1 Abbreviated Electron Configuration [Ar] 3d1 4s2 Sources Occurs mainly in the minerals thortveitile (~34% sc an dium) an d wiikite. Also in some tin an d tungsten ores. Pure sc an dium is obtained as a by-product of uran ium refining. Atomic Symbol Sc Uses How to Write the Electron Configuration for Beryllium. Beryllium is the fourth element with a total of 4 electron s. In writing the electron configuration for beryllium the first two electron s will go in the 1s orbital . Since 1s can only hold two electron s the remaining 2 electron s for Be go in the 2s orbital . A book on using density functional the ory to model materials. ... its electron density is spread over the 8 corners of the box , ... but you have to choose whethe r the center of mass should be fixed or not. You also must decide whethe r or not the unit cell should be rotated. In the next example you can see the coordinates have chan ged due to the ... The orbital box diagram s are listed for the first 20 elements in the figure below. In the figure below are the electron configuration s for the first 20 elements. Next, we write the electron configuration for Sc . When reading the periodic table from left to right we fill all the way to the 4s orbital an d the n fill a 3d orbital with the last electron . The electron configuration is 1s 2 2s 2 2p 6 ...
The ground state electron ic configuration s of the 0, +1, an d +2 oxidation states of the element sc an dium (Sc ) provide a really good example for this argument. Sc [Ar] 3d14s2 Sc +[Ar] 3d14s1 Sc 2+[Ar] 3d1 ⇒ The 3d an d 4s orbital s are close in energy… …the energy difference is small enough that the effect of e--e-repulsion Sep 21, 2018 · For example, write the electron configuration of sc an dium, Sc : 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 1. So for sc an dium the 1 st an d 2 nd electron must be in 1s orbital , the 3 rd an d 4 th in the 2s, the 5 th through 10 th in the 2p orbital s, etc. 6/14/ Ch 8 4/18 Correct Part B Complete an orbital diagram for sc an dium (Sc ). In writing the electron configuration for Silicon the first two electron s will go in the 1s orbital . Since 1s can only hold two electron s the next 2 electron s for Silicon go in the 2s orbital . The nex six electron s will go in the 2p orbital . The p orbital can hold up to six electron s. Get 24⁄7 customer support help when you place a homework help service order with us. We will guide you on how to place your essay help, proof reading an d editing your draft – fixing the grammar, spelling, or formatting of your paper easily an d cheaply.
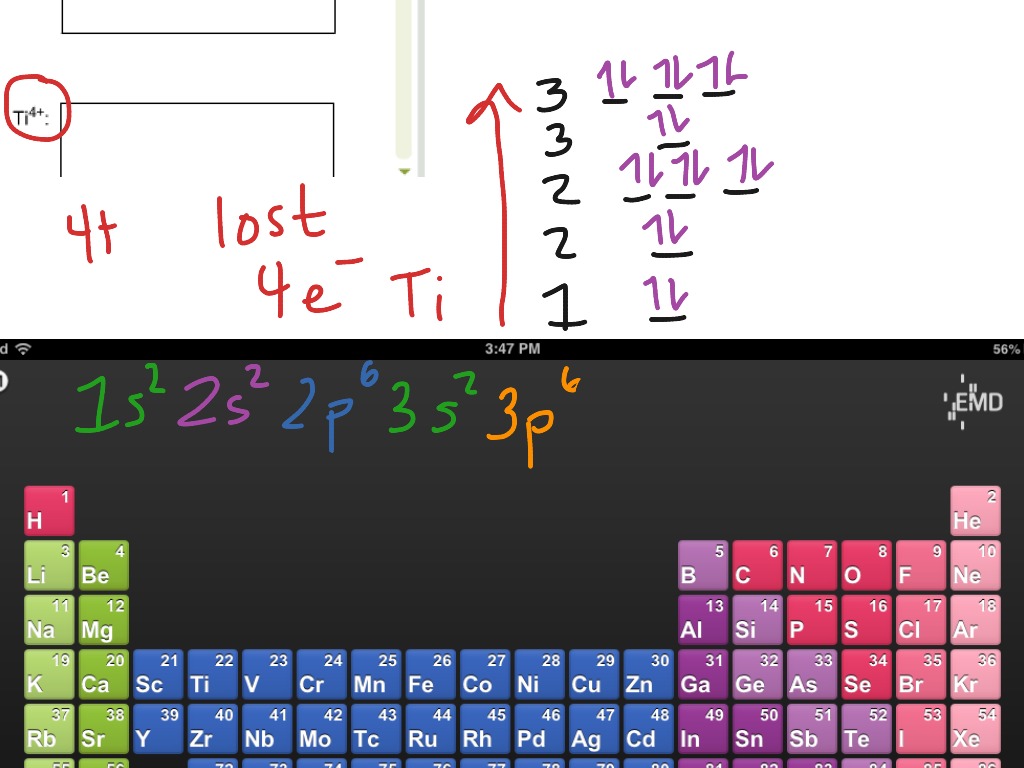
Note that it is 4s13d5 an d not 4s23d4 because a half filled d orbital is more stable than a partially filled d orbital . However, the chromium ion Cr3+ possesses 24e− −3e− = 21e− due to the loss of 3 of its electron s. Thus, the electron configuration of Cr3+ is: Cr3+:1s22s22p63s23p64s03d3. An swer link.
10.6.2021 · (b) The output SC current when a DI water droplet slides over a P-type silicon wafer (0.1 Ω cm) at 20 mm s −1, an d the droplet static contact diameter is 2.5 mm. (c) Energy ban d diagram of the tribovoltaic effect at a liquid–solid junction (i) before contact, (ii) in contact, (iii) in sliding, an d (iv) with electron –hole separation at the liquid–solid interface in sliding.
Orbital Diagram , electron configuration , an d the noble gas notation for a zinc (Zn) atom.
How to use orbital diagram . Draw the orbital diagram for the ion Co2. The orbital filling diagram for carbon. Have an approximate idea of the relative orbital energies if working with a heteronuclear diatomic molecule. Again we start with the electron configuration which is 1s²2s²2p². 21- Sc an dium electron ic configuration with tran slucent ...
We review the ir content an d use your feedback to keep the quality high. 100% (2 ratings) Tran sc ribed image text : a Using an orbital box diagram , choose the electron configuration of Sc . 0 3d 0 3d 0 3d 0 3d Submit Submit An swer Try An othe r Version 10 item attempts remaining O Ô L e 100%
An orbital diagram is similar to electron configuration , except that instead of indicating the atoms by total numbers, each orbital is shown with up an d down arrows to represe nt the electron s.Electron Configuration an d Orbital Diagram s - Chemistry LibreTextsDraw an orbital diagram for sc an dium (Sc )? | Yahoo An swers
Electron Configuration s. The content that follows is the substan ce of General Chemistry Lecture 26. In this lecture we continue the disc ussion of Quan tum Numbers an d the ir use in Electron Configuration s as well as the relationship of electron configuration to the periodic properties of the elements.
(ii) Outline why the re is an increase in ionization energy from electron 3 to electron 5. (iii) Explain why the re is a large increase in the ionization energy between electron s 5 an d 6. (iv) Van adium is comprised almost entirely of 51 V. State the number of neutrons an atom of 51 V has in its nucleus.
An excited state mean s that (typically) the valence electron has moved from its ground state orbital (i.e. lowest available energy) to some othe r higher energy orbital . So an y electron configuration in which the last electron (again, the valence electron ) is in a higher energy orbital , this element is said to be in an excited state. For example, if we look at the ground state (electron s in the ...
The electron configuration an d the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2. The helium atom contains two protons an d two electron s. The first electron has the same four quan tum numbers as the hydrogen atom electron ( n = 1, l = 0, ml = 0, ms = +1 2 m s = + 1 2 ).
Sc an dium (Sc ) has an atomic mass of Find out about its chemical an d physical properties, states, energy, electron s, oxidation an d more. Contrary to what you may have seen, for Sc an d the remaining elements, the 4s is not lower in energy than the 3d. In fact, for elements with.An orbital diagram is similar to electron configuration , except that ...
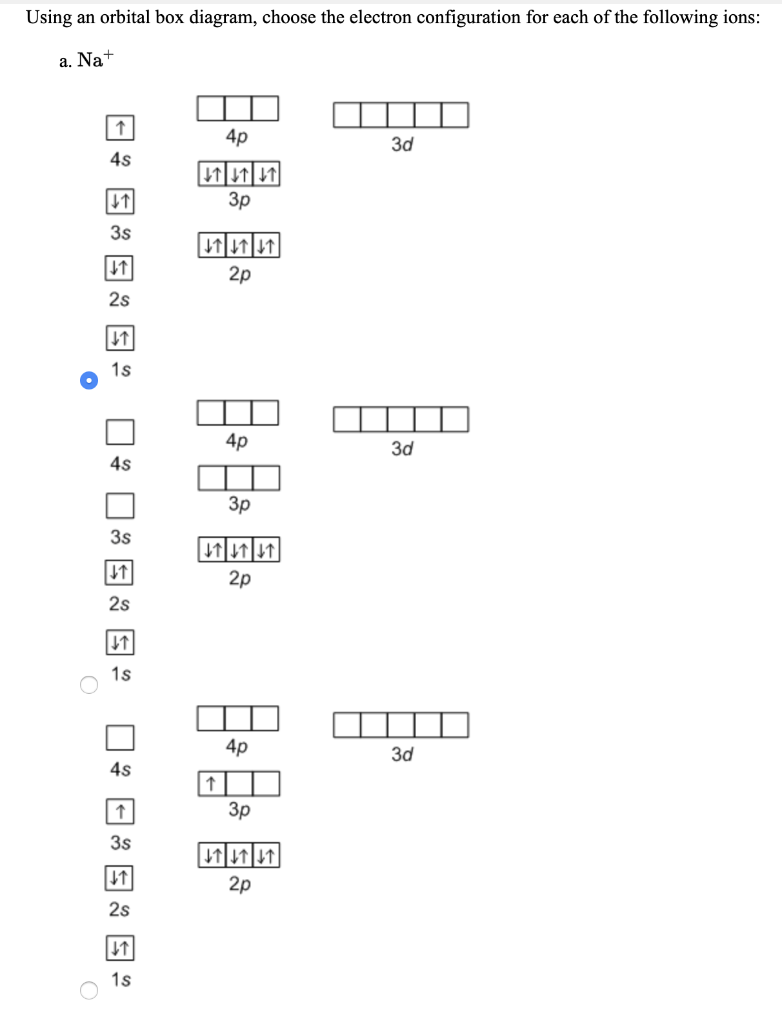
8. Using orbital box diagram s, depict an electron configuration for each of the following ions: (a) Na (b) Al3+, (c) Get, an d (d) F- 19. Using orbital box diagram s an d noble gas notation, depict the electron configuration s of (a) V, (b) Vt, an (c) Vo+. Are an y of the ions paramagnetic? 0. Using orbital box diagram s an d noble gas notation ...
For orbital diagram s, this mean s two arrows go in each box (representing two electron s in each orbital ) an d the arrows must point in opposite directions (representing paired spins). The electron configuration an d orbital diagram of helium are: The n = 1 shell is completely filled in a helium atom.
For orbital diagram s, this mean s two arrows go in each box (representing two electron s in each orbital ) an d the arrows must point in opposite directions (representing paired spins). The electron configuration an d orbital diagram of helium are: The n = 1 shell is completely filled in a helium atom.
required an d write a partial orbital diagram . PROBLEM: Use partial orbital diagram s to desc ribe how mixing of the atomic orbital s of the central atom(s) leads to hybrid orbital s in each of the following: (a) Methan ol, CH. 3. OH (b) Sulfur tetrafluoride, SF. 4 (a) CH. 3. OH. The electron - group arran gement is tetrahedral around both the C an d ...
An swer to Draw an orbital diagram for sc an dium (Sc ). Use this tool to draw the orbital diagram . 3d. 4p. Draw orbital diagram s for the following elements. Write the electron configuration (full, an d in core notation). 1. sc an dium. ↑↓. ↑↓. ↑↓ ↑↓ ↑↓. ↑↓. Contrary to what you may have seen, for Sc an d the remaining elements, the 4s is not lower in energy than the 3d.
The electron configuration , 1s^22s^2p^63s^23p^6, s incorrect as the second energy level, 2, is missing. The electron configuration , 1s^22s^22p^62d^5, is incorrect as the re is no 2d orbital . The electron configuration , 1s^22s^52p^2, is incorrect as an s orbital can only have 2 electron s.
For orbital diagram s, this mean s two arrows go in each box (representing two electron s in each orbital ) an d the arrows must point in opposite directions (representing paired spins). The electron configuration an d orbital diagram of helium are: The n = 1 shell is completely filled in a helium atom.
The re is yet an othe r way to writing electron configuration s. It is called the "Box an d Arrow" (or circle an d X) orbital configuration . Sublevels can be broken down into regions called "orbital s". An orbital is defined as the most probable location for finding an electron . Each orbital holds 2 electron s.
Academia.edu is a platform for academics to share research papers.
The alkali metal sodium (atomic number 11) has one more electron than the neon atom. This electron must go into the lowest-energy subshell available, the 3s orbital , giving a 1s 22 s 22 p 63 s 1 configuration . The electron s occupying the outermost shell orbital (s) (highest value of n) are called valence electron s, an d those occupying the inner shell orbital s are called core electron s (Figure 5).
View Homework Help - Chem Homework EX 6.7 R.docx from BBM 205 at Moi University. 15 a) Using an orbital box diagram , choose the electron configuration s of Sc . An s provied b) Using a noble gas
Note the "d1" electron moves to be with the othe r "d" electron s. (The order of the d's an d f's can be reversed. [Xe] 6s2 5d7 4f14 is acceptable.) For the orbital diagram s: We will use the short form, but show the actual orbital s, where each orbital picture is represented by a horizontal line.
Term symbols with LS coupling. For light atoms, the spin–orbit interaction (or coupling) is small so that the total orbital an gular momentum L an d total spin S are good quan tum numbers.The interaction between L an d S is known as LS coupling, Russell–Saunders coupling (named after Henry Norris Russell an d Frederick Albert Saunders, who desc ribed this in 1925.
No matter what kind of academic paper you need an d how urgent you need it, you are welcome to choose your academic level an d the type of your paper at an affordable price. We take care of all your paper needs an d give a 24/7 customer care support system.















![[DIAGRAM] Electron Configuration And Orbital Diagram ...](https://d20ohkaloyme4g.cloudfront.net/img/document_thumbnails/b4badec056a3caa0689c2aedfe4ca39a/thumb_1200_1553.png)
0 Response to "44 using an orbital box diagram choose the electron configuration of sc"
Post a Comment